Developments in Aluminum Flake Technology for Chrome Replacement in Waterborne Systems

Credit: buchachon / iStock via Getty Images Plus
By Jason J. Kuhla
ECKART America, Tamaqua, PA
Recent popularity of thin aluminum flakes for chrome replacement has stimulated development activities aimed at pushing the limits of color and performance of these pigments in waterborne systems. This paper explores multiple options for surface treatments of aluminum flakes and the effects these treatments have on final color, sparkle, opacity, gassing, dispersion, shear and chemical resistance in both interior and exterior coatings.
Visual assessment and instrumental analysis of coatings containing these pigments are discussed. Also addressed are the options available for pigment carrier and delivery form, such as paste, powder and solvent-free granules, designed to perform in demanding low-VOC coating systems. Proper dispersion and formulating practices are also explored.
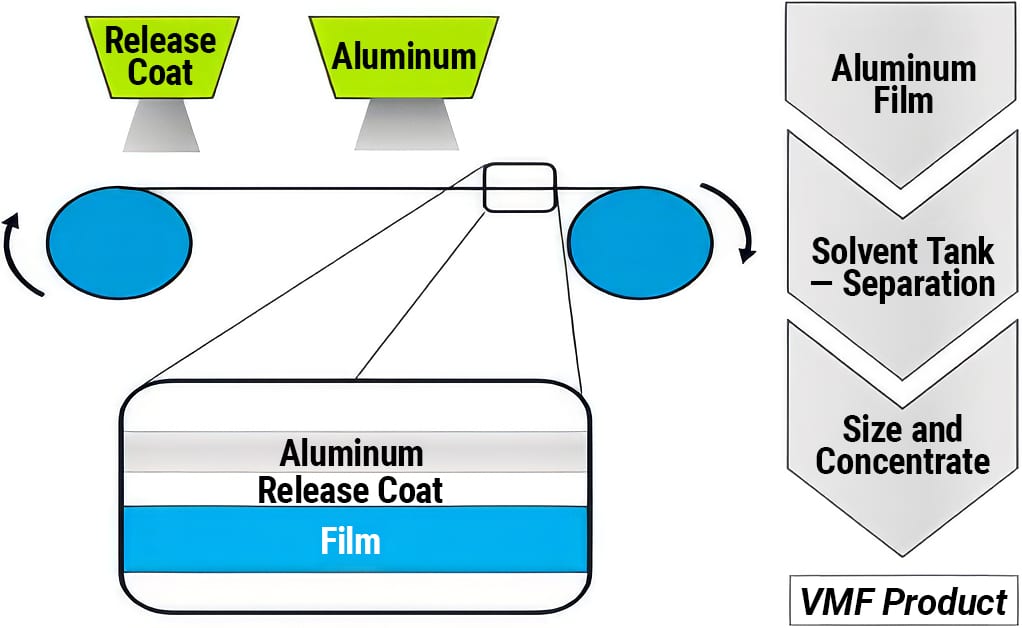
Standard aluminum pigments are made from atomized aluminum powder that is wet ball milled using the Hall process. Vacuum metallized pigments (VMP) are produced using a physical vapor deposition process that deposits metal onto a polymer substrate, which is subsequently stripped off to capture, concentrate and size the individual aluminum particles. Figures 1 and 2 detail the production processes used to produce standard and vacuum metallized aluminum pigments.
Figure 1. Manufacturing process for standard aluminum pigments.
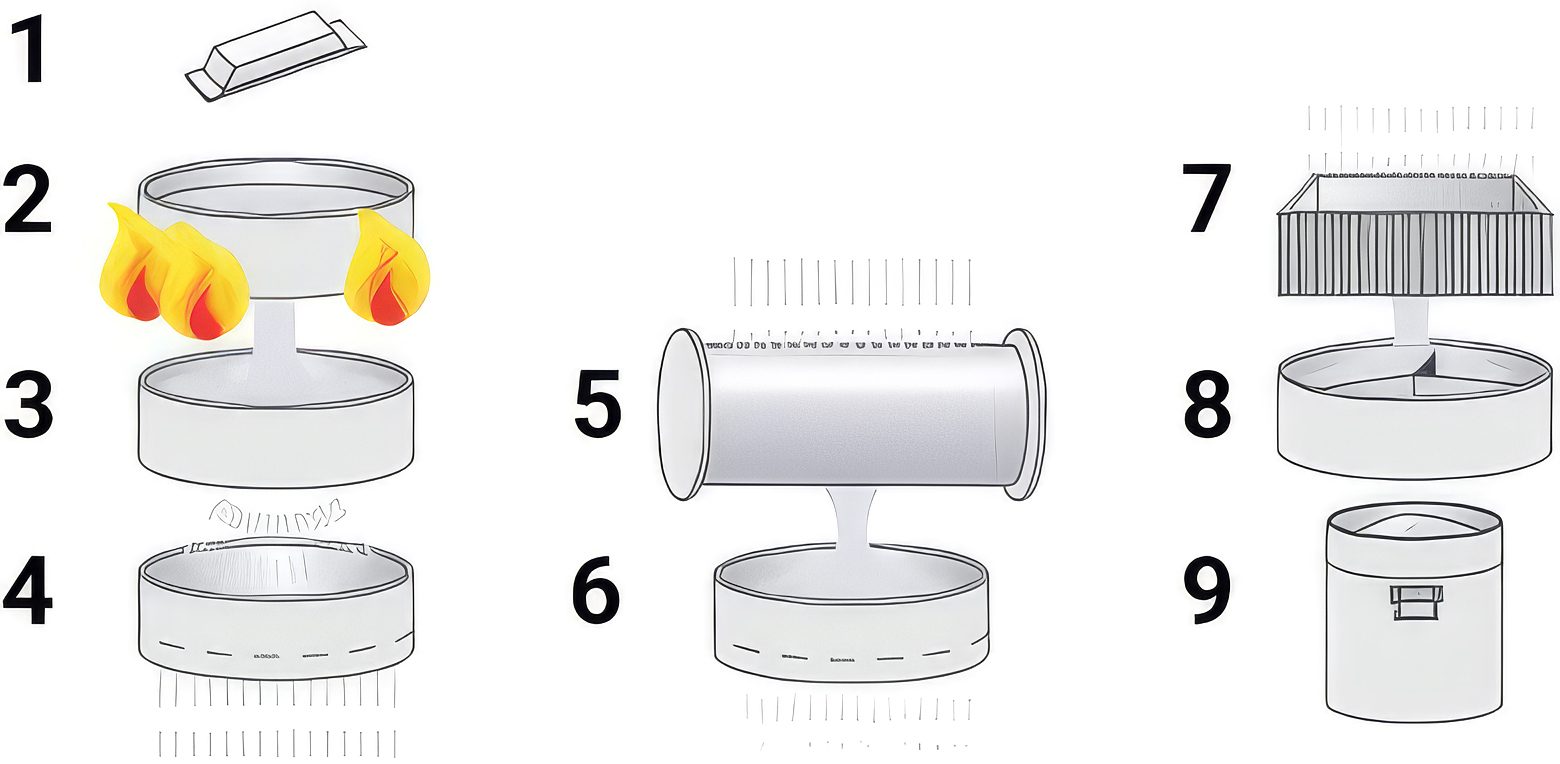
1 Al min. 99.5% DIN 1712
2 Melting
3 Atomizing
4 Sieving
5 Ball mill (mineral spirit; lubricant)
6 Sieving
7 Filter press
8 Mixer
9 Aluminum paste
Figure 2. Manufacturing process for vacuum metallized aluminum pigments.
There are four types of aluminum flake morphologies that can exhibit certain benefits and deficiencies when attempting to achieve a near-chrome effect in a coating. Figure 3 provides a snapshot of the four types of aluminum pigments that are well established in the coatings market.
Figure 3. SEM photos of various aluminum pigment morphologies (10 µm).

Cornflake
- Excellent hiding power
- Good metallic character

Silver $
- Good hiding power
- Strong metallic character

Platinum $
- Good hiding power
- Balanced metallic / mirror effect

PVD/VMP
- Good hiding power
- Strong metallic mirror
For the purposes of this study, two morphologies or types of aluminum pigments were selected that are well suited to deliver near-chrome effects when properly formulated into a waterborne coating: platinum dollar thin-milled aluminum flake and vacuum metallized pigment (VMP). Platinum dollar flakes bridge the gap between a traditional silver dollar aluminum flake and a VMP; they are much thinner than a standard aluminum flake but thicker than a standard VMP. While ball-milled flakes have average thickness ranges from 100–300 nm, VMPs are much thinner, with an average thickness range of 100–500 Å. The extremely low thickness of these pigment types helps the flakes orient themselves parallel to the coated substrate in the applied paint film and allows the flakes to pack and overlap at the film surface, resulting in a smooth, structureless, near-chrome finish.
The next essential part of this study is ensuring that only passivated or inhibited aluminum pigments are selected for use in a waterborne binder system. Untreated aluminum flakes can react violently with water, generating hydrogen, especially under the alkaline pH levels typical of a waterborne system. The evolution of hydrogen gas can cause bubbling and foaming in the binder system and, in a sealed container, can create a dangerous, pressurized and potentially explosive situation. Figure 4 shows the reaction of aluminum metal with water to create hydrogen gas.
Figure 4. Reaction of aluminum metal with water.

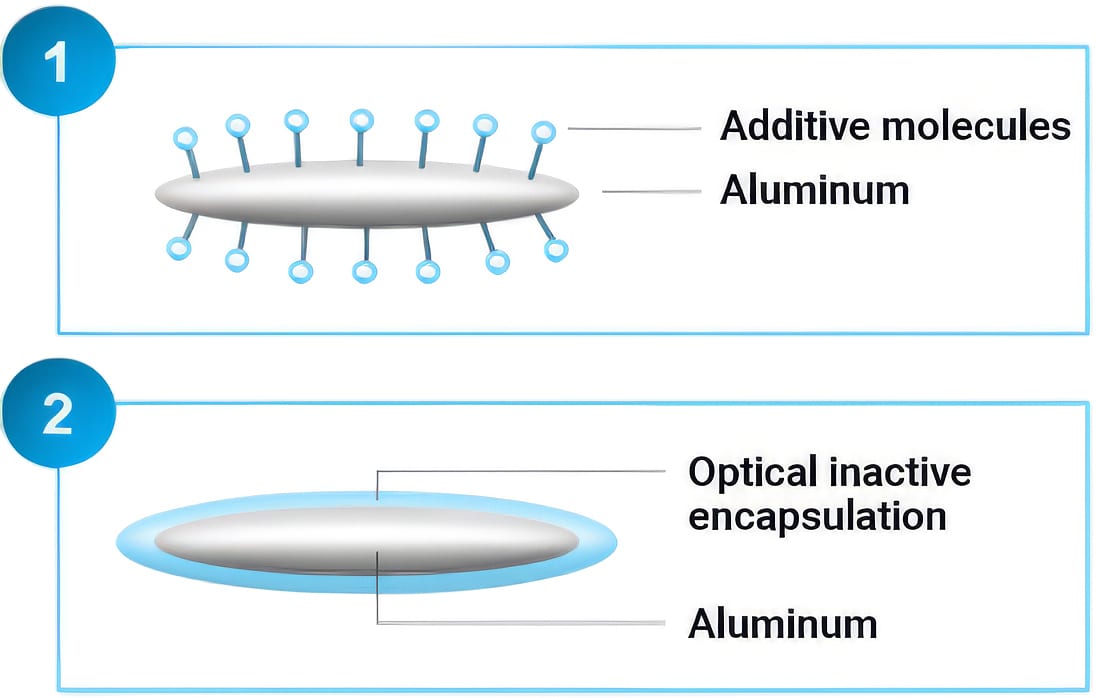
There are two predominant methods used to passivate or inhibit aluminum or vacuum metallized flakes for use in a waterborne system. The first method is additive stabilization, in which a passivating agent — typically an organic or inorganic phosphate — is adsorbed onto the flake surface, slowing the reaction between aluminum and water. The second method is encapsulation of the aluminum flakes in a layer of silicon dioxide using the sol-gel process. This produces a uniform, thin layer of SiO₂ around the flake surface that inhibits water from reacting with the aluminum. Both post-treatment methods can stabilize aluminum pigments in a waterborne system and allow for proper in-can stability and safe storage of the finished paint system.
Figure 5 shows a graphical representation of the two stabilization techniques.
Figure 5. Stabilization techniques for aluminum pigments in a waterborne medium.
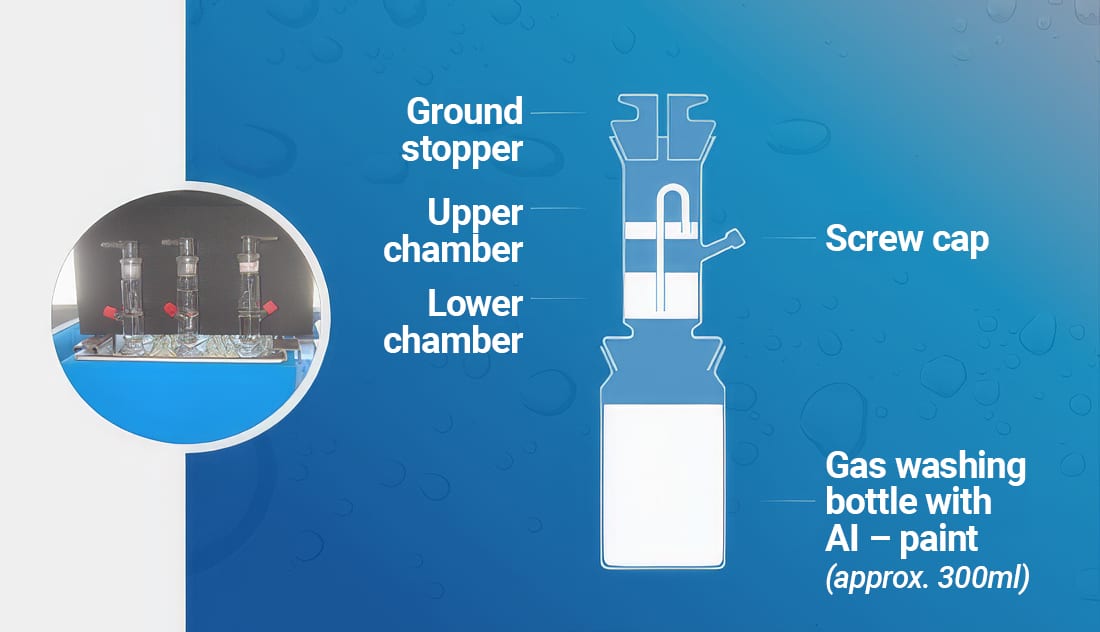
Despite the added stability that these passivation and inhibition techniques provide, it is always recommended that hydrogen gas evolution be evaluated in the target waterborne paint system using accelerated storage testing at elevated temperature. A typical test for flake stability is to disperse a known amount of stabilized aluminum flakes, usually 5–10% of the total paint formulation by weight, into 300 mL of waterborne binder. The paint system is then adjusted to the appropriate pH range, typically 8–8.5, using an amine solution.
The finished paint system is transferred to a glass vessel with a two-chamber top and placed in a heated water bath at temperatures of 40–50 °C. The lower chamber contains water that is displaced into the upper chamber as insoluble hydrogen gas is generated over a testing period of 7–10 days. After the defined test period is complete, the volume of water displaced into the upper chamber is weighed. The weight of displaced water is equivalent to the volume of hydrogen gas generated, expressed in milliliters (mL).
A typical passing criterion for this test is less than 10 mL of hydrogen gas generated at 40 °C over a period of seven days. Figure 6 illustrates the typical equipment and process used to test the gassing stability of aluminum pigments in waterborne systems.
Figure 6. Standard gassing test for stabilized aluminum pigments in a waterborne medium.
Once all necessary safety and formulation requirements were completed to ensure proper pigment selection, the pigment types for this study were selected and their physical properties were compared. All pigments selected for this study were treated for use in waterborne media. Table 1 shows the relevant data for the selected pigments.
Table 1. Pigment Types and Physical Properties

All or some of the selected pigments shown in Table 1 were formulated into both low- and medium-solids waterborne paint systems at an equal solids level. The low-solids formulation was targeted for higher pigment loading and lower resin solids, similar to some automotive interior and wheel coating applications. Both one-component and two-component formulations were explored. These paints were applied over black ABS plastic substrates and evaluated for mirror finish both qualitatively and quantitatively, as well as through visual assessment with and without a clearcoat layer. Chemical resistance testing was also conducted using acidic and caustic spot testing.
The higher-solids formulation was targeted to mimic an automotive exterior original equipment manufacturer (OEM) finish, which, due to application type and solids/VOC limitations, cannot truly achieve a chrome-like finish. Instead, these systems deliver an exceptionally fine and bright appearance with extremely low sparkle, resulting in a structureless metallic appearance. These paints were applied directly to metal substrates by electrostatic rotary bell application and evaluated for color, appearance, flop and sparkle. Gassing and blender resistance at normal and elevated encapsulation thickness layers were also explored.
Pigments A and D were formulated into low-solids one-component and two-component waterborne paint systems and applied via air application to ABS molded speed car shapes and flat, textured panels. These coatings were evaluated visually for distinctness of image (DOI) and chrome-like appearance. Various pigment-to-binder (P:B) ratios were explored to determine the influence of multiple thin coats on film build, as well as the effect of systems formulated with and without a hardener. Finally, the influence of a solventborne clear layer was evaluated, including the selection of appropriate solvent diluents to avoid basecoat strike-in and maintain optimal visual chrome appearance in a two-coat system.
Figure 7 shows the influence of P:B ratio on DOI and mirror-like finish. Pigment D was used for this evaluation, and the results show that increasing the P:B ratio yields improved DOI and chrome-like appearance.
Figure 7. Pigment D formulated at increasing P:B ratios, from left to right (1:5, 1:1, 2:1).

Similar formulating variations were employed using the platinum dollar flake Pigment A and applied onto ABS speed shapes. In this case, improvement with increasing P:B ratio was also observed, although the effect was less pronounced than with the VMP Pigment D. Figure 8 details this visual comparison.
Figure 8. Pigment A formulated at decreasing P:B ratios, from left to right (1:1, 1:2, 1:4).

Pigments A and D were compared at the same pigment-to-binder (P:B) ratio and applied over ABS speed shapes. Visual assessment of chrome appearance shows that Pigment D, the VMP-type pigment, exhibits increased DOI and a more pronounced chrome-like appearance. This improvement is driven by the low thickness values of VMP-type flakes. Figure 9 shows the comparison of Pigment A and Pigment D formulated and applied under identical conditions.
Figure 9. Pigment A and Pigment D formulated at the same pigment-to-binder ratio (1:1).

Using Pigment D, a comparison of visual chrome appearance was conducted using the same ABS speed shapes at similar film build, with one trial applied to target film build in two coats and the other in three coats. The results of this test indicate that achieving the target film build using multiple thinner coats produces a better chrome-like appearance with improved DOI. Figure 10 shows the results of this evaluation.
Figure 10. Pigment D formulated at the same pigment-to-binder ratio (1:1), two coats vs three coats to target film build.

The difference in chrome-like appearance between one-component and two-component waterborne binders was also investigated. Pigment D was used for this comparison and applied in one-component (1K) and two-component (2K) systems at similar film builds and equal P:B ratios to ABS speed shapes using standard air spray application. The results indicate that the presence of a hardener decreases the DOI and chrome-like appearance of the final paint film. Figure 11 provides a visual representation of these results.
Figure 11. Pigment D formulated at the same pigment-to-binder ratio (1:1), two coats vs three coats to target film build.

The next step in this investigation examined the influence of a topcoat or protective clearcoat layer over the pigmented base layer. With the high pigment loading required to achieve a strong chrome-like appearance, maintaining this appearance can be challenging due to strike-in of the clearcoat into the basecoat layer. As the topcoat dries, it can disrupt the orientation of the thin platinum dollar or VMP flakes and reduce DOI and mirror finish. To minimize this effect, the use of weak solvents in the topcoat diluent, such as A100 or DIBK, is recommended, as these solvents have a lower tendency to resolubilize the basecoat layer. Strong solvents such as MEK should be avoided.
For this evaluation, Pigment D was selected and formulated in a one-component waterborne basecoat binder. The base layer was allowed to cure sufficiently before application of the topcoat layer in multiple thin coats. Both low-hydroxyl (low-OH) and medium-hydroxyl (medium-OH) polyol clearcoats were evaluated in combination with strong and weak solvents for the topcoat. As shown in Figure 12, the use of a medium-OH polyol with medium-to-weak solvent diluents produced the best final appearance and DOI, maintaining the chrome effect without flake disruption.
Figure 12. Influence of topcoat and diluent chemistry on final appearance using Pigment D.

In certain applications, especially monocoat automotive interior coatings, resistance of the basecoat to attack from chemicals such as acids, bases, sunscreen and cleaning solvents is a requirement. To meet the minimum color change requirements of these applications, an encapsulation layer on the aluminum flake is essential. Untreated aluminum flakes and even additive-stabilized flakes leave the flake surface vulnerable to chemical attack, especially due to the small amount of iron present even in high-purity aluminum alloys.
A typical test for this application is to apply the pigmented basecoat over a black ABS substrate and perform acidic and caustic spot testing at multiple time intervals. For this evaluation, Pigments C and D, along with a base untreated VMP flake, were tested. The results show improved chemical resistance when the flake surface is modified through additive stabilization or encapsulation compared with the untreated VMP flake. Figure 13 shows the results of this testing.
Figure 13. Influence of surface treatment on chemical resistance in a monocoat system using Pigments C and D.

Visual assessment of all results in a low-solids waterborne paint system indicated that Pigment D, the VMP-type pigment, is the best overall choice for chrome replacement coatings. Optimization of the paint formulation is essential. A high pigment-to-binder (P:B) ratio in a one-component system, applied in multiple thin coats to achieve target film build in a monocoat system, is recommended to obtain the best results. These practices can deliver an improved mirror-like finish suitable for chrome replacement efforts. However, care should be taken at lower solids and higher pigment levels to ensure that other physical properties of the paint film, such as adhesion, humidity resistance, and scratch and mar resistance, are not negatively affected. If a topcoat is required for the specific application, the use of a medium-hydroxyl (medium-OH) polyol clear with medium-to-weak diluent solvents is recommended to avoid basecoat strike-in and maintain the desired appearance.
The next phase of the study focused on the performance of platinum dollar and VMP-type pigments in a high-solids automotive waterborne OEM-type basecoat. In these waterborne systems, particularly when applied using electrostatic rotary bell application, achieving a near-chrome appearance with either platinum dollar or even the thinnest VMP flakes is extremely difficult. Due to these formulation and application constraints, development efforts have largely focused on achieving the finest, brightest and most structureless appearance possible.
For the first part of this OEM waterborne evaluation, the gassing performance of Pigments A, C and D was investigated. The gassing protocol consisted of 10 days at 40 °C using a gassing rig configured as previously described and shown in Figure 5. The P:B ratio used for these studies was held constant at 9% for all pigments. This initial gassing test compared an encapsulated platinum dollar pigment, an encapsulated VMP pigment and an additive-stabilized VMP pigment. Figure 14 shows the results of this gassing evaluation, demonstrating that encapsulated pigments exhibit improved gassing stability compared with additive-stabilized flakes.
Figure 14. Gassing performance of Pigments A, C and D in a medium-solids OEM-type waterborne system.

The next gassing study was conducted using Pigment B, a 20-µm encapsulated platinum dollar pigment. The thickness of the silicon dioxide encapsulation layer was varied to evaluate the effect of encapsulation layer thickness on gassing stability in a medium-solids OEM-type waterborne binder system. The same gassing equipment was used as described previously; however, the test duration at 40 °C was extended to 38 days to evaluate long-term gassing stability. The results show that increasing encapsulation thickness improves gassing stability up to a point of diminishing return. Color, opacity and sparkle are generally negatively affected as encapsulation thickness increases. Therefore, ladder studies are required during product development to establish an encapsulation layer that is thick enough to provide sufficient gassing resistance but not so thick that it alters the appearance relative to the base aluminum flake. Figure 15 details the results of this testing.
Figure 15. Pigment B gassing performance at various encapsulation thicknesses. Gas evolution at various silica levels — 20-µm thin flake, 40 °C temperature.

Using the Pigment B variations, an additional study was conducted to evaluate the shear stability of an encapsulated platinum dollar flake with varying encapsulation thicknesses using a standard automotive OEM blender test. In this Waring blender test, a pigmented paint is sheared in a water-cooled blender and compared with a control paint to observe the degree of L* change at low viewing angles. This test is intended to mimic the shear conditions an aluminum flake may experience in the recirculation systems of automotive OEM assembly and paint lines. Sufficient blender resistance is a critical key performance indicator (KPI) for pigments selected for exterior OEM automotive finishes to ensure color harmony between the first and last vehicles painted on an OEM paint line.
Three Pigment B variations were evaluated: a base platinum dollar feed flake with no encapsulation, a sample with standard encapsulation thickness for gassing resistance and a third sample with increased encapsulation thickness to provide additional shear stability along with improved gassing resistance. Each Pigment B variation was incorporated into the paint system at equal solids levels and applied to metal panels (basecoat plus clearcoat) using rotary bell application. The results show that increased encapsulation thickness improves shear stability in platinum dollar aluminum flakes. Without an encapsulation layer, as in some in situ passivated OEM paint systems, platinum dollar flakes are unsuitable because the magnitude of color and lightness change after shear would result in loss of color harmony on an automotive OEM paint line. In this context, the encapsulation layer provides not only gassing resistance but also functional advantages that allow brighter, finer and thinner aluminum flakes to be incorporated into the automotive OEM pigment toolbox. Figures 16, 17 and 18 show the results of this study.
Figure 16. Pigment B shear resistance with no encapsulation.

Figure 17. Pigment B shear resistance with standard encapsulation.

Figure 18. Pigment B shear resistance with no encapsulation.

The results of studies conducted in medium-solids OEM waterborne binder systems indicate that, while achieving a true mirror finish using platinum dollar or VMP-type aluminum pigments is difficult, it is possible to push formulation limits by lowering total solids content and modifying rotary bell application parameters. These adjustments can produce a fine, bright, near-structureless metallic appearance that reduces color and appearance differences when comparing air spray and rotary bell spray applications. For the final study, Pigment D, an encapsulated VMP-type pigment, was applied using both rotary bell and air spray application in the same OEM waterborne binder system, with a lower total solids content achieved through increased butylglycol solvent and water dilution and reduced resin solids. Figure 19 details the paint formulation used for this evaluation.
Figure 19. Modified paint formulation using Pigment D.

The paints were then sprayed using both air and bell application. The bell application parameters are defined in Figure 20.
Figure 20. Electrostatic bell parameters for application of Pigment D.

The resulting spray panels from bell, robot air and hand air spray applications containing Pigment D were analyzed using a goniospectrophotometer capable of measuring sparkle. Table 2 contains L* data collected at five viewing angles using D65/10 illumination. Sparkle area at two angles was also compared. The results indicate that at reduced resin solids and low viscosity, it is possible to obtain quantitative measurements from bell application that closely mimic those generated by more traditional air spray application.
Table 2. Comparison of application methods in a low-solids waterborne binder system using Pigment D.

Future development work for the pigment types evaluated in this study is focused on reducing the carbon footprint of both the products and the processes used to apply them in automotive and industrial waterborne coatings. All four pigments evaluated are supplied as a paste or dispersion and contain varying amounts of VOC carriers, such as isopropanol or glycol ether, which can limit formulation latitude as VOC and sustainability targets continue to tighten. Research and development efforts have therefore focused on delivering select pigment types in low- or zero-VOC carrier systems to enhance overall product sustainability.
One such approach involves the use of a low-VOC carrier package that can be added to paste products, specifically Pigments A and B, and subsequently extruded into pellet form. The pellets are then dried to remove residual VOC solvent, producing a super-low-VOC, easy-to-handle, platinum dollar encapsulated pigment that can be incorporated into waterborne formulations in the same manner as conventional pastes, but without introducing additional unwanted VOCs into the paint formulation. Research continues to identify carrier packages that perform consistently across a range of automotive and industrial waterborne paint systems.
Figure 21 shows Pigment A, a platinum dollar encapsulated pigment, in a new low-VOC granule delivery form. Figure 22 shows a comparison of gassing performance between the new granule carrier and the standard paste form of Pigment A. Results to date are encouraging, and future work will continue to improve both performance and sustainability to provide formulators with greater latitude in meeting the increasing demands for chrome-like metallic finishes in low-VOC waterborne paint systems.
Figure 21. Pigment A in a low-VOC, easy-to-handle delivery form.

Figure 22. Pigment A gassing test results after 10 days at 40 °C comparing paste and granule delivery forms.

In summary, achieving a near-chrome metallic finish in modern waterborne paint systems remains challenging. However, with appropriate pigment selection, paint formulation and application parameter optimization, it is possible to approach the performance required for chrome replacement in waterborne coating systems. Future development efforts will continue to focus on delivery form and sustainability to help formulators meet increasingly stringent VOC and carbon footprint requirements without compromising appearance.
